
What if we could reboot sick cells?
Key findings:
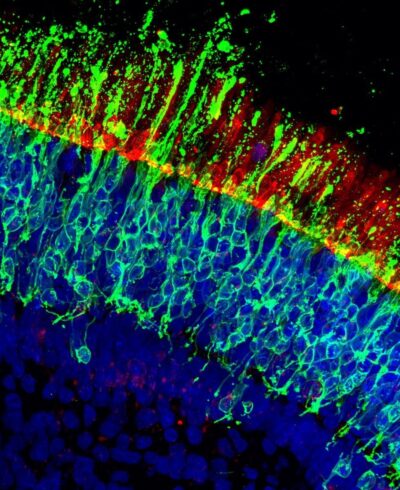
- MitoCatch directs donor mitochondria to specific cell types, including neurons, retinal, cardiac, endothelial, and immune cells.
- Binder-mediated mitochondrial delivery increases uptake compared to untargeted methods.
- Donor mitochondria internalized via MitoCatch become cytosol-exposed, are dynamic, and capable of fusion / fission within recipient cells.
- Protein binders can be engineered to tune delivery efficiency and specificity.
- Targeted transplantation improved survival of damaged neurons in vitro and retinal ganglion cells in vivo, and was well tolerated in animal models, with no detectable immune response.
Mitochondrial dysfunction contributes to numerous currently untreatable conditions, including neurodegenerative disorders such as Parkinson’s and Alzheimer’s, optic nerve atrophy, and heart failure. While transplantation of healthy mitochondria has been explored as a potential therapy, conventional methods lack the specificity and efficiency needed to reach disease-affected cells.
MitoCatch overcomes these limitations by using engineered protein binders to link donor mitochondria to target cells. The system employs three complementary strategies: binders on the cell surface (MitoCatch-C), binders on mitochondria (MitoCatch-M), and bispecific binders bridging mitochondria and cell membranes (MitoCatch-Bi). By tuning binder affinity and multivalent interactions, researchers achieved efficient, cell type-specific mitochondrial delivery in human and mouse models across multiple cell types.
“MitoCatch represents a paradigm shift in how we think about treating mitochondrial diseases. The versatility of the system is particularly exciting: whether targeting neurons in the brain, photoreceptors in the eye, or cardiac cells in the heart, MitoCatch can be adapted to deliver healthy mitochondria precisely where they are needed most, opening new possibilities for diseases that currently have no cure.”
TEMURKHAN AYUPOV & VERONICA MORENO-JUAN, co-first authors of the paper
“Our vision is to advance this technology into a therapy that can restore cellular health and function in patients affected by these devastating diseases.”
BOTOND ROSKA, last author
Scientific contact:
Botond Roska, Head of the Human Retinal and Central Visual Circuits Group
E-Mail: headoffice_roska@iob.ch
Illustrations: © IOB, 2026, all rights reserved
Original Publication
Cell type-targeted mitochondrial transplantation rescues cell degeneration
Temurkhan Ayupov, Verónica Moreno-Juan, Serena Curtoni, Alex Fratzl, Upnishad Sharma, Susana Posada-Céspedes, Ramona Ratiu, Rei Morikawa, Alexandra Graff Meyer, Margherita Pezzoli, Glenn Bantug, Morgan Chevalier, Yanyan Hou, Sarah A. Nadeau, Álvaro Herrero-Navarro, Vikram Ayinampudi, Elizabeth Kastanaki, Natasha Whitehead, Rebecca A. Siwicki, Mariana M. Ribeiro, Ji Hoon Han, Annalisa Bucci, Christoph Hess, Simone Picelli, Magdalena Renner, Daniel J. Müller, Cameron S. Cowan, Simon Hansen, Botond Roska
Nature. 2026 April 15.
doi: 10.1038/s41586-026-10391-0
Scientific releases:
Media releases:
Recent News